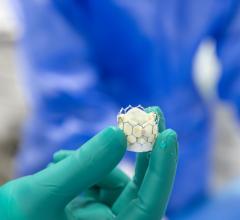
June 7, 2024 — Medtronic today announced new data from the CoreValve Evolut Clinical Program, reinforcing the positive ...
Structural Heart
This structural heart channel includes news, videos, podcasts and other content related to diagnosis and treatment of structural heart disease. Topics covered include heart valve repair and replacement, transcatheter aortic valve replacement (TAVR), transcatheter mitral valve replacement (TMVR), transcatheter tricuspid valve replacement (TTVR), left atrial appendage (LAA) occlusion, heart failure interventional device therapies, and closing holes in the heart using, including occlusion of atrial septal defects (ASDs), ventricular septal defects (VSDs) and patent foramen ovales (PFOs).
June 5, 2024 — Edwards Lifesciences announced it has entered into a definitive agreement to sell its Critical Care ...

Here is a look at the Top 10 pieces of content viewers were reading during the month of May.
1. American College of ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
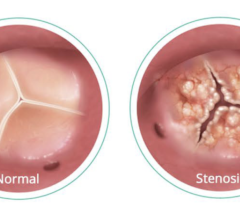
May 15, 2024 — A new study demonstrated parity between a minimally invasive procedure to replace the aortic valve in the ...
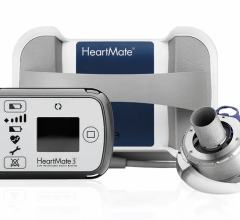
May 15, 2024 — The U.S. Food and Drug Administration (FDA) announced that Abbott is recalling the HeartMate 3 LVAS by ...
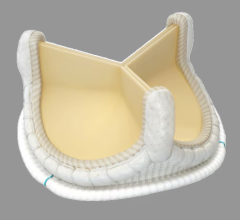
May 8, 2024 — 4C Medical Technologies, Inc. ("4C Medical"), a medical device company dedicated to advancing minimally ...
May 7, 2024 — Medtronic announced the release of important clinical outcomes in two leading transcatheter valve ...
May 6, 2024 — Teleflex Incorporate, a leading global provider of medical technologies, today announced that the Wattson ...

Clinical trials and innovative technology took center stage during the month of April, racking up some record number ...
April 25, 2024 —Heart-Valve-Surgery.com, a leading patient advocacy group for heart valve disease, with support from Med ...
April 25, 2024 — Atlantic Health System’s Morristown Medical Center treated the first patient in New Jersey using Edward ...
April 23, 2024 — Medtronic plc, a global leader in healthcare technology, today announced the launch of its latest ...
April 17, 2024 —CPR Therapeutics, Inc. (CPR-T), an early-stage medtech startup funded by the N.I.H and N.S.F to develop ...
April 16, 2024 — Vivasure Medical, a company pioneering novel fully absorbable technology for percutaneous vessel ...
April 9, 2024 — UC Davis Health cardiology team members are among the first in the country to treat patients with tricus ...


 June 07, 2024
June 07, 2024