A comprehensive review or more than 80 randomized controlled trials (RCTs) investigating how to best manage optimal ...
Venous Therapies
This channel includes news and new technology innovations of Venous Therapies including deep vein thrombosis (DVT) and vena cava filters.
February 22, 2021 — The U.S. Food and Drug Administration (FDA) recently cleared the Cook Medical Zilver Vena Venous ...
October 26, 2020 — The U.S. Food and Drug Administration (FDA) has cleared the Medtronic Abre venous self-expanding ...
September 9, 2020 — The Society of Interventional Radiology (SIR) published new clinical practice guidelines that ...
July 20, 2020 – BD (Becton, Dickinson and Company) recently completed the acquisition of Straub Medical AG, a privately ...
October 31, 2019 – In a large series of iliac vein stent cases, a blinded comparison found intravascular ultrasound ...
August 13, 2019 — A simple change in the way health professionals track their patients’ progress has brought improved ...
May 17, 2019 — The U.S. Food and Drug Administration (FDA) this week approved Fragmin (dalteparin sodium) injection, for ...
May 6, 2019 — The U.S Food and Drug Administration (FDA) has cleared the Boston Scientific Vici Venous Stent System for ...
April 3, 2019 — The U.S. Food and Drug Administration (FDA) recently cleared Bard Peripheral Vascular's Venovo Venous ...
February 12, 2019 — A study presented at the 2018 annual meeting of the Cardiovascular and Interventional Radiology ...

Catheter-based blood clot removal a decade ago was a standard of care for acute coronary revascularization, but declined ...
August 8, 2018 — Boston Scientific Corp. announced it has signed an agreement to acquire Veniti Inc., which has ...

The introduction of thermal ablation revolutionized the treatment of varicose veins, yet recurrence remains a stubborn ...
April 10, 2018 – Cardiva Medical announced the company received U.S. Food and Drug Administration (FDA) approval for an ...

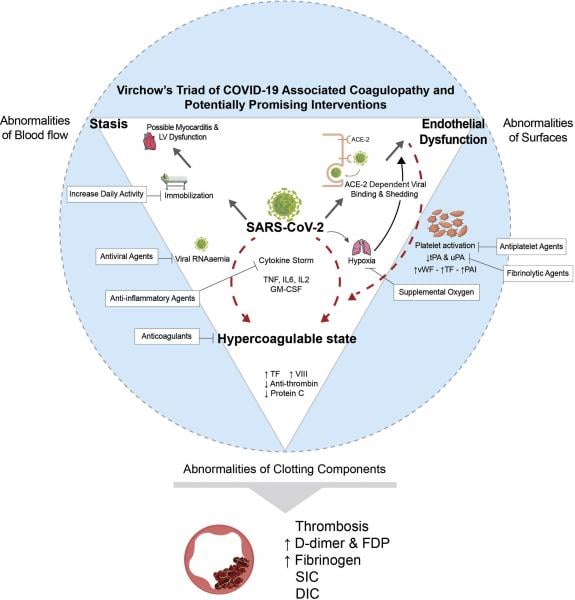
 March 22, 2021
March 22, 2021