This is an example of artificial intelligence automation for cardiac MRI using an AI app from TeraRecon's Envoy AI marketplace. The GE MRI image has been countoured, anatomy labeled and all quantification automated by the AI, greatly reducing post-processing time. See this and other new AI imaging technologies from the 2019 RSNA meeting.
The key question I am always asked at cardiology conferences is what are the trends and interesting new technologies I have seen. Based on these observations and looking ahead into 2020, here is my list of hottest cardiac technologies I am watching. These will likely play a larger role in clinical practice in the near future. I included links to example articles and some of the videos we shot regarding each topic so you can see the technologies discussed.
1. Artificial Intelligence in Cardiology
By far, the No. 1 overarching hot topic at all the medical conferences I attended over the past couple years is artificial intelligence (AI), and its newest iteration in the form of machine learning (ML). What was once science fiction or far-fetched research projects are now starting to gain U.S. Food and Drug Administration (FDA) market clearance. Some AI elements are already being used without clinicians knowing it, being integrated into the backend of cardiology and radiology IT and reporting systems to help speed work.
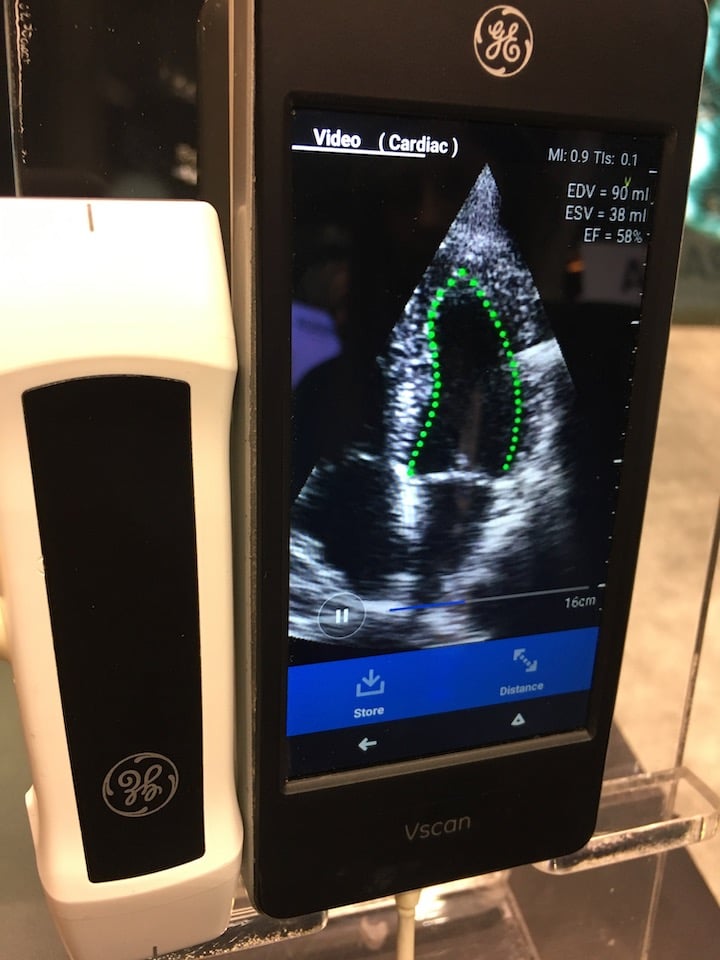
Dia artificial intelligence software performs an auto ejection fraction measurement on a cardiac ultrasound image from a GE Healthcare Vscan POCUS system. Photo by Dave Fornell
AI is helping augment cardiologists and medical imaging. Examples of AI commercially available today include automated ejection fraction (EF) calculations for point-of-care ultrasound systems (POCUS), like on the GE Healthcare Vscan. Premium cardiac ultrasound systems like the Philips Epiq use AI to automatically identify the anatomy, segment, label it, identify the optimal echo views and perform automatic measurements before the physician begins to read the case. Several vendors now offer AI-automated calcium scoring software for cardiac CT scans, creating the report quantification information in seconds and color coding the calcium by vessel segment on the dataset slices. Arterys AI-based cardiac MRI analysis software automates the quantification required to speed exam post processing. AI algorithms are being used to automatically detect arrythmias and send alerts to patients using wearables or smartphone-based apps that record ECG. Examples of this technology are the Apple Watch and the Kardia Alivecor device. AI will likely see its biggest steps forward in cardiology for point-of-care (POC) triage apps and wearables cardiac monitoring technologies. This will speed the process of getting at-risk patients examined by a human cardiology specialist and aid in earlier detection of cardiovascular diseases.
VIDEO: ACC Efforts to Advance Evidence-based Implementation of AI in Cardiovascular Care — Interview with John Rumsfeld, M.D.
VIDEO: Artificial Intelligence to Automatically Risk Stratify Atrial Fibrillation Patients — Interview with Sanjaya Gupta, M.D.
VIDEO: Example of Artificial Intelligence Integrated Into Cardiac Ultrasound
VIDEO: Overview of Artificial Intelligence and its Use in Cardiology — Interview with Anthony Chang, M.D.
2.Transcatheter Mitral and Tricuspid Valve Interventions
In 2019, the FDA cleared the use of transcatheter aortic valve replacement (TAVR) for all patient surgical risk categories, the final step to opening the procedure to all patients and making it a new standard of care. Clinical study data shows TAVR is as good as, or better, than surgical valve replacements, and is being performed as a same-day out patient procedure at some centers. Experts predict 75 percent of aortic valve replacements will be performed via TAVR by 2025.
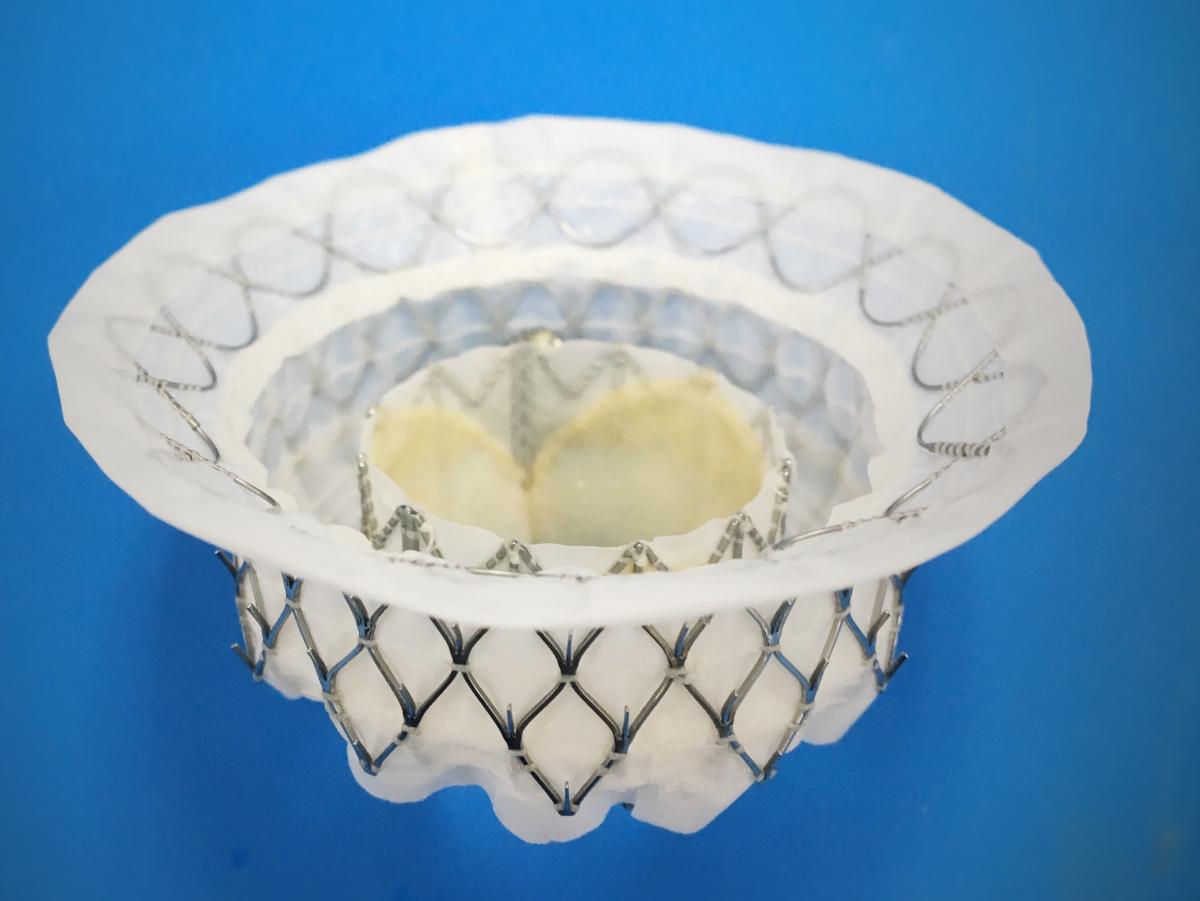
The Medtronic Intrepid TMVR system is currently enrolling patients in the pivotal APOLLO Trial. The study plans to analyze the safety and efficacy of the Intrepid system in up to 1,200 patients with severe, symptomatic MR. The results are expected no earlier than 2021.
With the success of TAVR, cardiologists attention has rapidly shifted toward transcatheter mitral valve repair and replacements (TMVR) and transcatheter tricuspid valve repair and replacements (TTVR). Several devices in both of these spaces are working their way through international and U.S. FDA trials. The big three interventional device vendors, Medtronic, Abbott and Boston Scientific, along with Edwards Lifesciences, all have made massive investments in TMVR and TTVR technologies, including purchasing several start-up valve companies.
These will likely be the hottest structural heart topics at the interventional meetings in 2020.
VIDEO: The Expansion of TAVR Following the FDA Clearing its Use in All Patients — Interview with Torsten Vahl, M.D.
Developments in Transcatheter Mitral Valve Replacement
TriClip Device For Tricuspid Regurgitation Effective at One Year in TRILUMINATE Study
3. Wearable Technologies Aid Patient Monitoring
Nearly everyone now carries a smartphone, and millions of people use wearable devices to track their health using Apple Watch or Fitbit devices. So, it should not be surprising that these devices and smartphone apps are being leveraged by both consumers and healthcare professionals to better monitor patient health. Instead of an annual visit, health tracking information for many patients is now available 24-7 for the entire year. Smart software and AI is being incorporated into the wearable and app algorithms to detect abnormally high heart rates, arrhythmias, and other factors to alert patients to contact their doctors. This consumer-grade early warning network will likely play a big role in the coming years to automatically triage patients and help know when they should seek professional healthcare help.

An example of a wearable cardiac tracking device offered by the vendor Sky Lab displayed at the American Heart Association (AHA) 2018 meeting. It is worn like a normal ring on a finger and interfaces with a smartphone app via a wireless Bluetooth connection to track ECG readings and alert a patient if an arrhythmia is detected. Photo by Dave Fornell.
A large number of healthcare systems are integrating wearables into their remote patient monitoring programs. In some cases, professional grade wearables, like inexpensive stick-on ECG monitors now offered by several vendors, are replacing traditional Holter monitors. Some hospitals are using wearables and consumer-grade devices like glucose monitors, wireless scales and blood pressure monitors that interface with smartphone apps to track cardiac rehabilitation, and diabetic and heart failure patients remotely.
VIDEO: Use of Wearable Medical Devices for Cardiac Rehabilitation — Interview with Robert Klempfner, M.D.
VIDEO: Use of Wearables to Track Electrophysiology Patients — Interview with Khaldoun Tarakji, M.D.
VIDEO: The Future of Wearables in Healthcare — Interview with Karl Poterack, M.D.
4. Virtual and Augmented Realty in Cardiology
Augmented reality (AR) and virtual reality (VR) are now being used by several large cardiology device vendors for staff training. Boston Scientific is using VR headsets with pre-loaded live electrophysiology device implant cases, which offer a 360 degree view of the EP lab and the procedure as a key opinion leader offers his advice and explains what they are doing during the case. This virtual physician proctoring may help speed up the transfer of how-to knowledge and patient access to new EP technologies. Abbott uses a virtual reality simulator to train interventional cardiologists how to use its intravascular optical coherence tomography (OCT) imaging technologies to exam at lesions in the coronary arteries, or create 3-D renderings of the disease vessel segments. The training in the VR headset puts the operators into a virtual cath lab with a 360 degree of the room and equipment to perform the imaging.
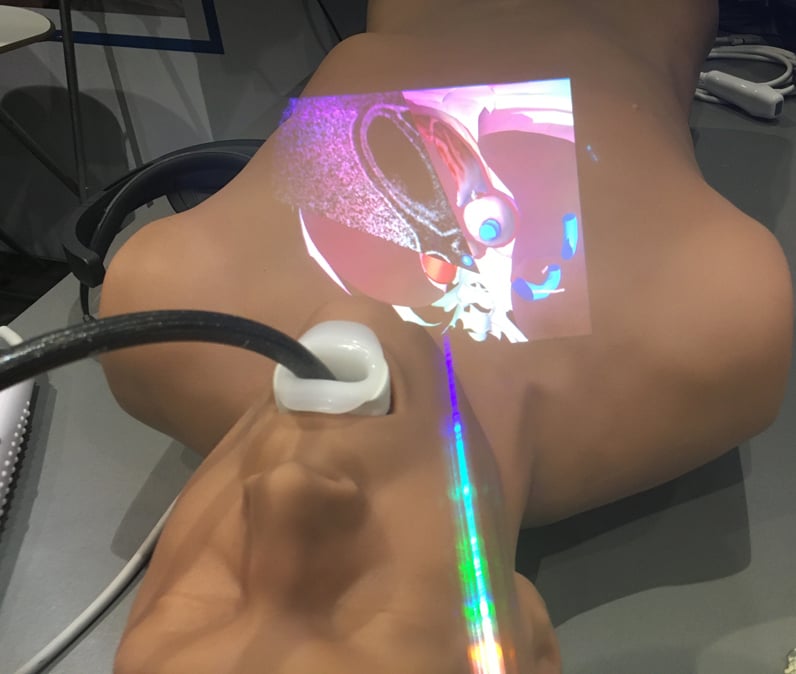
Augmented reality being used for transesophageal echo (TEE) cardiac imaging training displayed by CAE at RSNA 2019. Photo by Dave Fornell
NovaRad has FDA clearance on the first surgical AR system. The surgeon wears an augmented reality headset and they can superimpose and co-register a CT or MRI dataset onto a patient on the OR table. They can then virtually slice through the patient to pre-plan the procedure and mark their skin for incisions based on being able to to the underlying tissues.
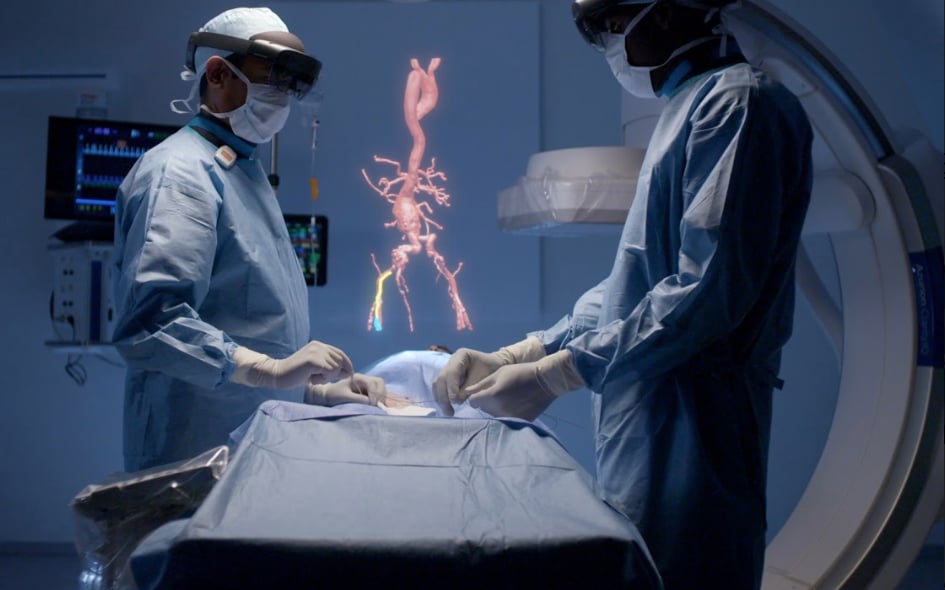
With the use of the Microsoft Hololens, Philips is developing an augmented reality system to aid procedural guidance in the cath lab. In this photo, operators can see a 3-D hologram projected above the patient showing an aortic aneurysm and the femoral access route to implant a stent graft. The operators can rotate or enlarge the image using voice commands or hand movements. Photo from Philips Healthcare.
Philips Healthcare is a couple years into developing a cath lab augmented reality system. It enables an interventional radiologist or cardiologist to use hand movements or voice commands without breaking the sterile field. They can call up live procedural ultrasound in their AR visor, review CT imaging datasets they can slice through in mid-air, or 3-D holograms of the anatomy from rotational angiography or CT scans. These 3-D images in mid-air above the patient can be rotated and sliced through with hand movements to help with procedural navigation beyond what a 2-D overhead display screen offers. See an example of this technology this VIDEO.
VIDEO: Use of Virtual Reality to Aid Congenital Heart Disease — Interview with David M. Axelrod, M.D.
VIDEO: Augmented Reality Aids TEE Training By Showing Anatomy and Ultrasound Orientation
VIDEO: Users Can Touch This Virtual Reality Heart
5. Big Data and Structured Reporting
In the past decade of U.S. healthcare reform, the biggest accomplishment so far was to get the majority of hospitals to use electronic medical records (EMR) and digital reporting systems. While the government roadmapped computerized order entry and clinical decision support to be the next major efforts, healthcare systems have realized they can immediately leverage their EMR investments to data mine their rapidly growing digital archive. Analytics software are now the main priority for most hospitals and subspecialty departments. Data analytics from structured reports are being used to find workflow bottlenecks, monitor staff workloads, track numbers of procedures, disease types, inventory, patient outcomes, tracking costs, equipment usage, etc. What took hours to pull numbers for in Crystal Reports a few years ago can now be automated and questions answered in seconds. It is a new, more efficient way to manage staff, patient care and the medical business.
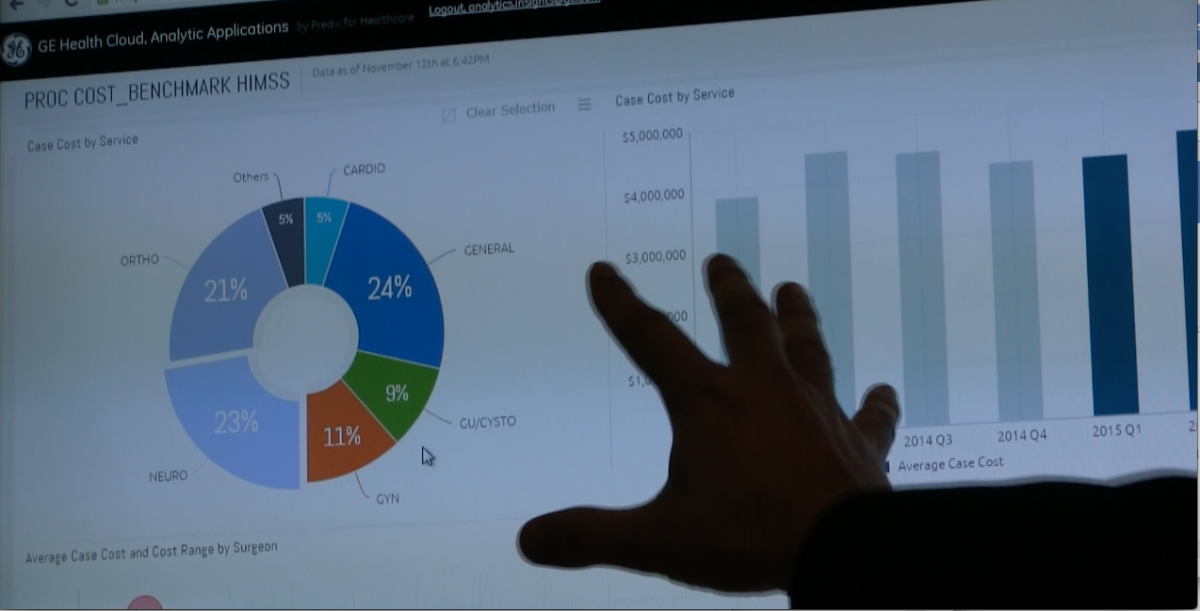
An example of analytics of patient EMR data being used to determine the actual costs of each type of surgical procedure being performed at a hospital using GE Healthcare's analytics software. Photo by Dave Fornell
The advanced next steps for mining this big data though is population health, where risk factors for all patient records in a health system can be automatically combed through to identify patients at risk for certain diseases that should be screened, such as atrial fibrillation or heart failure. We are at a point where physicians can reach out to patients for screenings to find early developing diseases so they can be prevented and managed, rather than the reactionary system used now to treat the result of end-stage diseases.
VIDEO: Minneapolis Heart Institute Saves With Analytics Software
Advanced Analytics Software for Cardiology
6. Holographic Procedural Navigation in the Cath Lab
In addition to augmented reality, live, 3-D, transesophageal echo (TEE) holographic imaging can be projected in the cath lab now without the need for special glasses or AR visors. At the TCT 2019 interventional meeting in September 2019, EchoPixel showed how a GE TEE system can project holograms of live views of heart valves or the left atrial appendage (LAA) using a special display screen. A couple large angiography system vendors are looking at the system for possible integrations with their own technologies. It may soon be possible to direct LAA occlusions or MitraClip implants using holograms and eliminate the need to have three different 2-D echo views so the operators can reconstruct the 3-D image in their brain. The holograms will make visualization much easier and intuitive for device deployments.
![]()
Echopixel created a technology that allows live transesophageal echo (TEE) 3-D imaging to be converted into live holograms on a special screen that does not require glasses. The photo shows an operator using hand movements to turn and slice through a dynamic image of a heart valve at the 2019 Transcatheter Cardiovascular Therapeutics (TCT) conference. Photo by Dave Fornell.
7. Robots in the Cath Lab
Robotic catheter navigation systems for both the electrophysiology (EP) lab and the cardiac cath lab have been around for several years now, but these systems are not common. The main questions limiting wider adoption is if these systems can help improve outcomes enough to justify the costs for the systems. But, another consideration for these systems is the fact that they allow remote control manipulation of catheters, which takes the operator out of the radiation field and allows them to take off their heavy lead aprons and take a seat in a control cockpit. This has appeal as procedures become more complex and longer duration, exposing operators to much more scatter radiation than in the past. As concerns about cumulative radiation dose in operators increases, these systems offer an alternative.
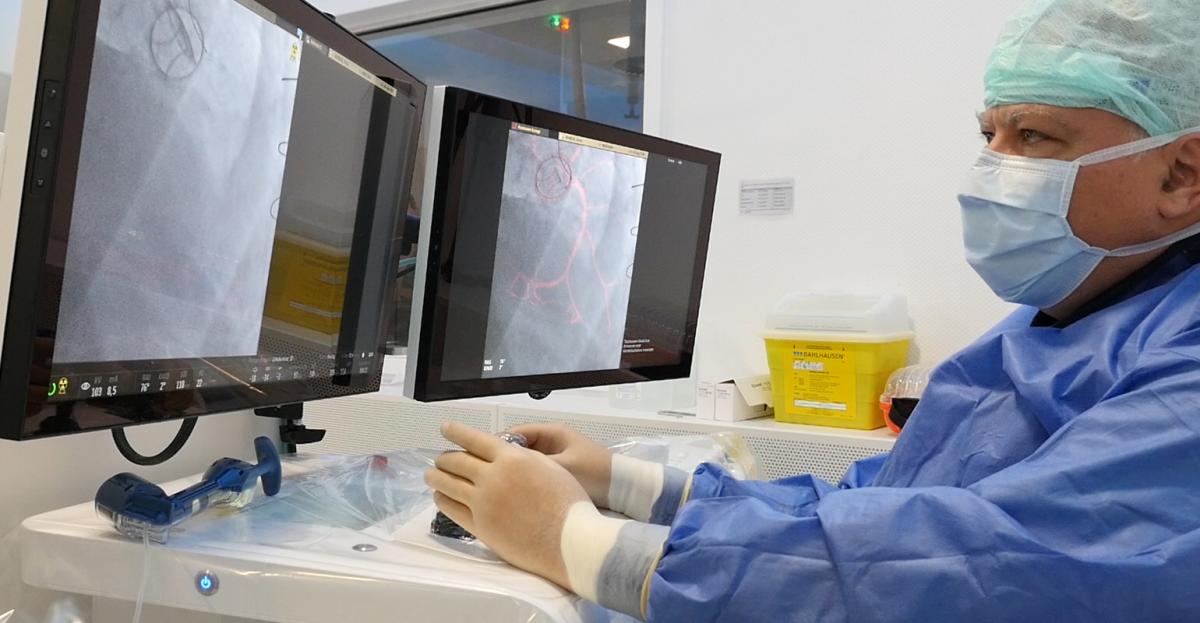
The Robocath R-One robot being used to perform PCI by Professor Michael Haude, director of Medical Clinic I at Rheinland Klinikum Neuss Lukaskrankenhaus, in Germany in December 2019. It has European CE mark clearance for PCI, and its first procedures were performed at centers in France and Germany in the fall of 2019. This system, and the Corindus CorPath system, both use a lead-lined cockpit in the cath lab for the operator to perform the procedure outside of the radiation field, sitting down and without the need to wear lead protection. Photo courtesy of Robocath.
The Corindus CorPath system was cleared by the FDA for use in straight forward percutaneous coronary interventions (PCI) and peripheral vascular cases. However, its real value might be with much more complex cases like chronic total occlusions (CTO). The system allows very precise, controlled millimeter increment movements.
Corindus was purchased in 2019 by Siemens Healthineers, which hopes to combine the robot into complete cath lab installation packages. Siemens also plans to expand the company's position for interventional radiology and non-cardiac vascular interventions. CorPath has European CE mark for neuro interventions and Siemens wants to expand this indication to new markets. Siemens said the CorPath performed its first neuro coiling case for an aneurysm in Canada in November 2019.
Another robotic contender is French-based Robocath. The company has European CE mark clearance for PCI, and its first procedures were performed at centers in France and Germany in the fall of 2019.
VIDEO: First Robotic PCI Case in Germany With Robocath R-One System
8. Virtual, Image-based FFR Technologies
A key diagnostic tool to determine if a stent is needed in a coronary lesion in the cath lab is pressure wire based fractional flow reserve (FFR). However, the technology requires the use of pharmacological stress agent adenosine and a wire pull back in the vessel, taking additional procedure time and adding cost to the procedure limiting adoption. Expanded use of FFR would likely take place if the technology was less expensive and easier to use. Two newer FDA cleared technologies offer image-based virtual FFR assessments — angiography-based FFR (FFR-angio) and computed tomography-based FFR (FFR-CT).
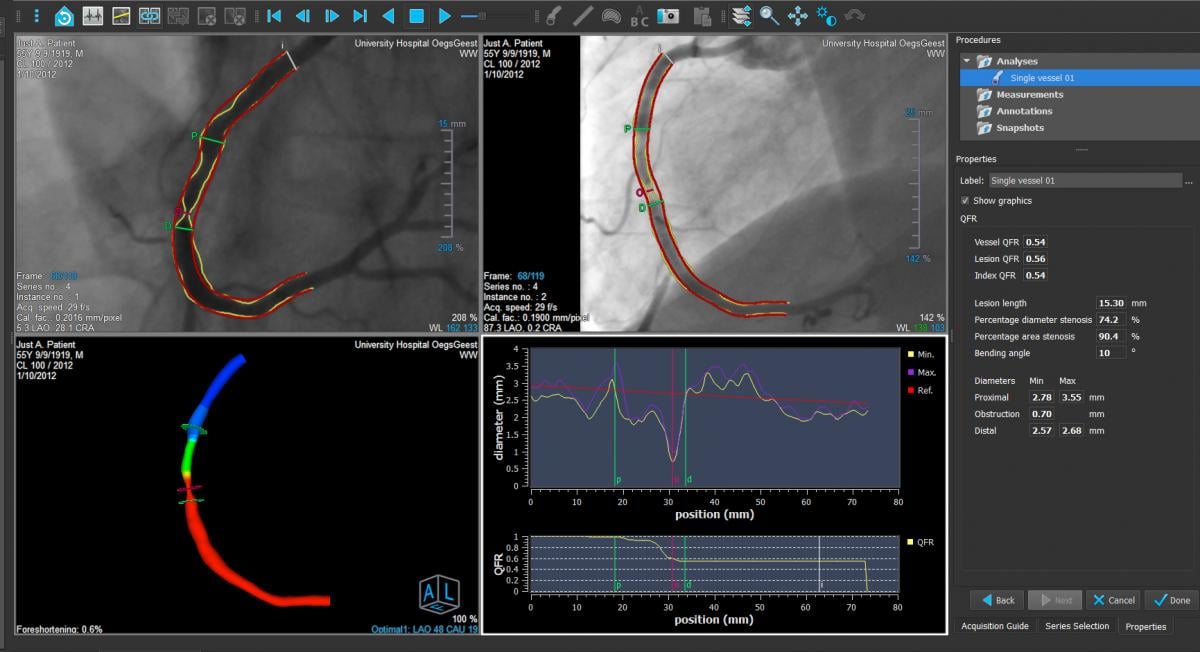
The is the FDA-cleared Medis FFR-Angio QCA software. It uses rotational angiography image acquisitions while the patient is on the table in the cath lab to analyze coronary vessel segments for a derived FFR ratio without the use of pressure wires. It also creates a 3-D reconstruction of the vessel showing a color code for the FFR values to help determine if a stent is needed and where is should be placed. Photo courtesy of Medis.
FFR-CT required the CT imaging to be sent to the vendor and it is run through a computational fluid dynamics algorithm and a supercomputer to create a 3-D coronary tree with FFR ratios for all the vessel segments. It can take a couple hours to process the results, but is fast enough to be used in some luminary hospitals to assess patients presenting with chest pain. FFR-CT is also starting to be used at some centers to help plan procedures.
The most recent iteration of this technology allows the user to virtually stent a vessel segment to determine the FFR value if the vessel was opened to its native lumen size. This is another planning tool to assess patients prior to entering the cath lab.
Another technology that two vendors now have FDA approval for is FFR-angio. This uses a rotational angiography image acquisition around the patient on the table and the software creates a 3-D coronary vessel reconstruction with a color coded showing of the FFR ratio. This can be performed table-side in the lab during a procedure and only requires a contrast injection and imaging.
VIDEO: Will CT-FFR Replace Diagnostic Angiograms? — Interview with Patrick Serruys, M.D.
VIDEO: Using FFR-CT in Everyday Practice — Interview with Kavitha Chinnaiyan, M.D.
I am always interested in readers' thoughts on new technologies and what they feel will be game-changers in the next decade. Feel free to drop me your thoughts at [email protected].
Related Cardiovascular Technology Advances Content:
Cleveland Clinic Unveils Top 10 Medical Innovations for 2020
The Future of Cardiology: 17 Technologies to Watch
Artificial Intelligence Can Detect Low-glucose Levels via ECG Without Fingerpick
A 40,000 Foot View of Trends in Cardiology
New Technology Highlights on the ACC 2019 Exhibit Floor
6 Key Health Information Technology Trends at HIMSS 2019
Echocardiography Trends at ASE 2019
7 Hot Topics in Cardiac CT Imaging
Key Technology and News at AHA 2018
6 Hot Topics in Interventional Cardiology at TCT 2019
VIDEO: Editor's Choice of the Most Innovative New Cardiac Technology at AHA 2018



 September 24, 2025
September 24, 2025