October 28, 2016 – Tryton Medical Inc., a primary developer of stents to treat coronary bifurcation lesions, and
...
This channel includes news and new technology innovations for stents, also called vascular scaffolds. Stents are used to help prop open a vessel treated by balloon angioplasty because of the barotrauma caused by the extreme stretching of vessel walls. The stent enables to vessel to heal in an open position with collapsing. Drug eluting stents (DES) are coated in anti-proliferative drugs to precent scar tissue growth which can cause restenosis and occlude the vessel. DES require antiplatelet therapy because the drug carrier polymer on DES can cause thrombus inside the stent, even years after treatment, which is why bare metal stents are still used in some patients. This page includes news on coronary stents, carotid stents, peripheral stents, bioresorbable stents, and renal stents.
...
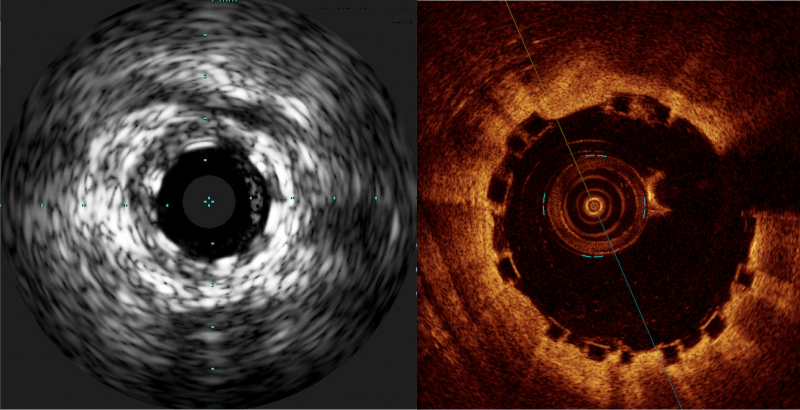
There has been a lot of interest in the interventional community regarding the Abbott Absorb Bioresorbable Vascular ...
October 13, 2016 — A surveillance project to evaluate the safety and effectiveness of transcarotid artery ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...

September 28, 2016 — The Cardiovascular Research Foundation (CRF) included 11 late-breaking trials and 16 first report ...
September 12, 2016 — Veniti Inc. has closed on $25 million in Series D equity financing from Boston Scientific Corp. The ...
August 24, 2016 — Intact Vascular Inc. announced that the one-year results from its Tack Optimized Balloon Angioplasty ...
There was an explosion of interest in bioresorbable stents immediately following the July 5, 2016 announcement that the ...

August 5, 2016 — Here are the top 20 most popular current content on the Diagnostic and Interventional Cardiology ...
August 2, 2016 —The U.S. Food and Drug Administration (FDA) granted market clearance for W. L. Gore & Associates’ Gore ...
July 26, 2016 — On July 20, Massoud Leesar, M.D., of University of Alabama at Birmingham Hospital implanted a patient ...
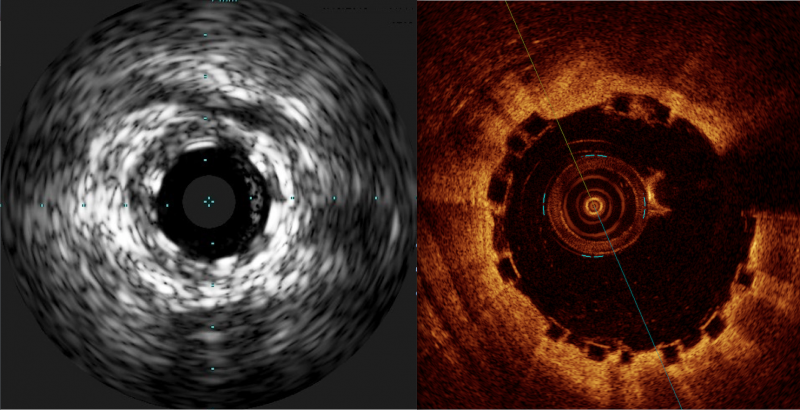
Some have labeled bioresorbable scaffolds (BRS), also known as bioresorbable stents, as the fourth evolution of ...
July 19, 2016 — Good Samaritan Hospital, Los Angeles, is the first hospital on the West Coast to offer patients with ...
July 11, 2016 — University Hospitals Case Medical Center, Cleveland, is among the first in the country – and the first ...
July 5, 2016 — The U.S. Food and Drug Administration (FDA) has cleared the first fully bioresorbable coronary stent for ...
June 6, 2016 — Amaranth Medical announced that it completed enrollment in May in the RENASCENT-II study of its novel ...