August 29, 2017 — C.R. Bard Inc. announced the Lutonix 035 Drug Coated Balloon PTA Catheter (DCB) has been granted ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
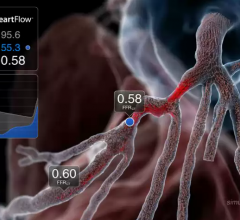
August 28, 2017 — Philips Healthcare and HeartFlow Inc. announced they entered into a collaboration agreement to improve ...
August 23, 2017 — PinnacleHealth is the first hospital in Pennsylvania and one of the first 10 in the country to ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
The Centers for Medicare and Medicaid Services (CMS) in August announced a proposed rule to cancel the Episode Payment ...
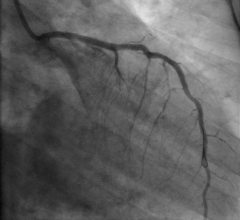
August 16, 2017 — A study has examined the efficacies of various post-percutaneous coronary intervention (PCI) ...

August 16, 2017 — The Centers for Medicare and Medicaid Services (CMS) announced a proposed rule to reduce the number of ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
August 15, 2017 — Surmodics Inc. announced receipt of an investigational device exemption (IDE) from the U.S. Food and ...
Aug. 15, 2017 – Patients undergoing percutaneous coronary intervention (PCI) at safety-net hospitals experienced similar ...
Aug. 15, 2017 — The U.S. Food and Drug Administration (FDA) has approved the Vascular Dynamics Inc. (VDI) ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
August 9, 2017 — HeartFlow Inc. announced that four Blue Cross Blue Shield companies have each issued a positive medical ...
August 9, 2017 — Ra Medical Systems announced that the United States Patent and Trademark Office (USPTO) has granted the ...
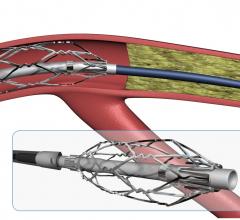
August 9, 2017 — Roxwood Medical Inc. recently announced it has entered into an exclusive agreement with Abbott for ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
August 4, 2017 — In petri dishes in her campus laboratory at New Jersey Institute of Technology, Alice Lee is developing ...
August 3, 2017 — Abbott recently announced the first patient has been enrolled in a clinical study evaluating the short ...

The current generation of hemodynamic monitoring systems can help catheterization labs electronically document the ...

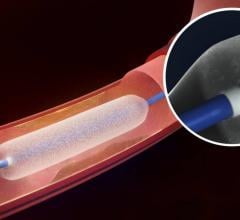
 August 29, 2017
August 29, 2017