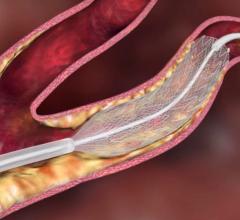
October 20, 2021 — Boston Scientific Corporation announced positive data for the Eluvia Drug-Eluting Vascular Stent ...
Stents
This channel includes news and new technology innovations for stents, also called vascular scaffolds. Stents are used to help prop open a vessel treated by balloon angioplasty because of the barotrauma caused by the extreme stretching of vessel walls. The stent enables to vessel to heal in an open position with collapsing. Drug eluting stents (DES) are coated in anti-proliferative drugs to precent scar tissue growth which can cause restenosis and occlude the vessel. DES require antiplatelet therapy because the drug carrier polymer on DES can cause thrombus inside the stent, even years after treatment, which is why bare metal stents are still used in some patients. This page includes news on coronary stents, carotid stents, peripheral stents, bioresorbable stents, and renal stents.
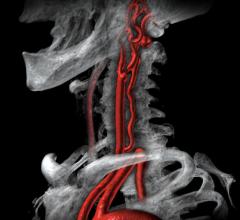
October 15, 2021 — The InspireMD CGuard Embolic Prevention Stent System (EPS) device for the treatment of carotid artery ...
October 4, 2021 — One month of dual antiplatelet therapy (DAPT) following stent implantation in high bleeding risk ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
September 7, 2021 — Carotid artery surgery and stenting have comparable long-term effects on fatal or disabling stroke i ...
September 1, 2021 — The STOPDAPT-2 ACS trial does not support the use of one month of dual antiplatelet therapy (DAPT) ...
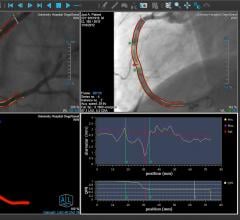
July 22, 2021 — Medis Medical Imaging is partnering with CORRIB Core Lab and Sinomed in randomized clinical trial of ...
June 30, 2021 — Abbott announced its Xience family of drug-eluting coronary stents received U.S. Food and Drug ...
June 7, 2021 — A couple years ago a study showed a mortality safety signal in patients who underwent peripheral artery ...
May 20, 2021 — Boston Scientific Corporation announced it has initiated the AGENT IDE trial for the Agent Drug-Coated ...
May 12, 2021 — Preliminary results of a clinical trial, presented today at the AATS 101st Annual Meeting, showed that a ...
May 1, 2021 - An analysis of a new international registry reveals benefits of using a longer covered stent for ...
April, 14, 2021 – Elixir Medical recently announced the first patient was treated in the BIOADAPTOR randomized ...
April 6, 2021 — Abbott today announced its Xience stent has received CE mark in Europe for shorter duration of dual anti ...
February 22, 2021 — The U.S. Food and Drug Administration (FDA) recently cleared the Cook Medical Zilver Vena Venous ...
February 9, 2021 — InspireMD Inc., developer of the CGuard Embolic Prevention System (EPS) for the prevention of stroke ...


 October 20, 2021
October 20, 2021




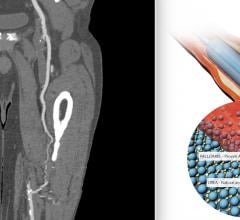




![Comparison showing platelet adhesion to the surface of various coronary artery drug-eluting stents (DES) in a preclinical study that used aspirin only. Abbott said the Xience stent's fluoropolymer is significantly more anti-thrombotic than other DES.[2]](/sites/default/files/styles/content_feed_medium/public/DES_Comparison_thrombus_formation_Stents_Abbott.jpg?itok=mfh9GUz-)

