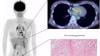
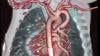
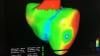
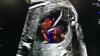
Dee Dee Wang, M.D., director of structural heart imaging, Henry Ford Hospital, Detroit, Mich., explains how patient survival depends on keeping the left ventricular outflow track (LVOT) clear and using 3-D imaging to predict what the neo-LVOT will look like prior to transcatheter mitral valve replacement (TMVR) procedures. The close proximity between the aortic and mitral valves in the left ventricle anatomy makes it critical to assess any mitral valve overhang that will obstruct blood flow out of the left ventricle. This issue has been raised in several cardiovascular imaging structural heart intervention planning sessions at conferences over the past two years, most notably at the Society of Cardiovascular Computed Tomography (SCCT).
Read the related article Interventional Imagers: The Conductors of the Heart Team Orchestra, which Wang helped author.
Watch the related VIDEO: Overview of the Henry Ford Hospital Structural Heart Program.
Additional articles and videos on Henry Ford Hospital