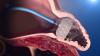
The Zoll LifeVest is a temporary, wearable defibrillator designed as a safety net for patients, especially those being evaluated for a permanent implantable cardioverter defibrillator (ICD). The Vest Prevention of Early Sudden Death Trial (VEST), presented at ACC 2018, was the first randomized, controlled, multi-center trial of the wearable cardioverter defibrillator. It looked to see if this device could effectively reduce sudden death in patients who had recently suffered a heart attack and had reduced heart function. Read more about the VEST Trial.
Videos
VIDEO: 3-Year Follow-up of the SYMPLICITY HTN-3 Trial
At TCT 2022, Deepak Bhatt, MD, MPh, Executive Director of Interventional Cardiovascular Programs, Brigham and Women's Hospital, presented a late-breaker session on Long-Term Outcomes Following Catheter-Based Renal Denervation in Patients With Uncontrolled Hypertensions: 3-Year Follow-up of the SYMPLICITY HTN-3 Trial. He spoke with DAIC editors about the outcomes.
Related TCT 2022 Coverage:
VIDEO: Unique Features of Coronary Artery Disease in Women
Cardiovascular Research Foundation’s (CRF) TCT 2022 in Review
Photo Gallery Highlighting TCT 2022
Cardiovascular Research Foundation’s (CRF) TCT 2022 in Review
TCT 2022 Honors Recipient of Thomas J. Linnemeier Young Investigator Award
TCT 2022 Announces Winner of Shark Tank Innovation Award
Educational Leaders Join Forces on Educational Opportunities
Top TCT 2022 Award Recipients announced by Cardiovascular Research Foundation (CRF)
EP Lab
Take a video tour of some of the medical devices designed to improve patient care, improve patient engagement and increase physiologic monitoring highlighted at the 2018 Consumer Electronics Show (CES). One of the most interesting technologies is a flexible electronics ECG monitor that can bend and twist with the skin and interfaces with a smartphone app. The exhibition includes more than 3,000 vendors and more than 170,000 attendees. For more examples of future healthcare technologies, watch the VIDEO “Editor's Choice of Future Healthcare Technologies at HIMSS.”
Emanuel Kanal, M.D., director of MRI services and professor of radiology and neuroradiology at the University of Pittsburgh Medical Center, explains the new mobile application he developed, which lets users visually model the forces at work during an MRI exam on patients with implanted medical devices, at the 2017 Radiological Society of North America (RSNA) meeting.
Read the related article “The Changing Relationship Between MRIs and Pacemakers.”
Vivek Reddy, M.D., director of cardiac arrhythmia services and professor of medicine, cardiology, Mount Sinai Hospital, N.Y., explains the latest Watchman left atrial appendage (LAA) occluder study data presented at the 2017 Transcatheter Cardiovascular Therapeutics (TCT) conference.
Watch the VIDEO “Post-FDA Approval Experience of LAA Occluders,” with David Holmes, M.D.
Watch a VIDEO interview with Reddy at Heart Rhythm Society (HRS) 2017 on leadless pacing technologies.
This video, provided by Acutus Medical, demonstrates a patient case showing the use of the AcQMap high-resolution electrophysiology (EP) mapping system. The system was cleared by the U.S. Food and Drug Administration (FDA) in October 2017. Read about the FDA approval. See more about this system in the VIDEO "Editor's Choice of Most Innovative New Technology at HRS 2017."
Leyla Elif Sade, M.D., MESC, professor of cardiology at Başkent University, Ankara, Turkey, discusses use of echo for cardiac resychronization therapy (CRT) lead optimization at the 2017 American Society of Echocardiography (ASE) annual meeting.
Related Content:
VIDEO Echocardiography Strain Fundamentals Made Easy
A Glimpse Into the Future of Cardiac Ultrasound
Andrea Natale, M.D., FACC, FHRS, FESC, executive medical director, Texas Cardiac Arrhythmia Institute at St. David's Medical Center, discusses how to build an electrophysiology (EP) program at the 2017 Heart Rhythm Society (HRS) annual meeting.
He explains what centers need to consider when creating an EP program, including what is required for device programs that include pacemakers, implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy (CRT), as well as the technologies and know-how for catheter ablation programs. He discusses who should be involved in creating an EP program and how EP needs to interface with other departments and subspecialties around the hospital.
Natale is a world recognized leader in the field of electrophysiology. Prior to the establishment of Texas Cardiac Arrhythmia Institute at St. David’s Medical Center, Natale was a member of the Cardiovascular Medicine Department at the Cleveland Clinic from 1999 to 2007, and served as section head for the Department of Cardiac Pacing and Electrophysiology and as medical director for the Cleveland Clinic’s Center for Atrial Fibrillation. In 2006, Dr. Natale was named to the U.S. Food and Drug Administration (FDA) Task Force on Atrial Fibrillation.
Read the article "Growing a Cardiac Rhythm Program in a Community Setting."
Lucas Boersma, M.D., Ph.D., FESC, St. Antonius Ziekenhuis, Nieuwegein, The Netherlands, discusses how subcutaneous implantable cardioverter defibrillator (S-ICD) technology works, selecting patient appropriate patients and new S-ICD technologies in development at the 2017 Heart Rhythm Society annual meeting. Read the article "Subcutaneous ICD System Shows Positive Outcomes in Largest Real-world Study."
Watch the related VIDEO: How to Install a Subcutaneous ICD System
Read the related article New Technology and Market Challenges Facing Implantable Cardioverter Defibrillators.
DAIC Editor Dave Fornell takes a tour of some of the most innovative new electrophysiology (EP) technology at the 2017 Heart Rhythm Society (HRS) annual meeting. This included new ablation and electromapping systems to improve cardiac ablation effectiveness, new wearable monitoring technologies that are replacing traditional Holter monitors, implantable cardiac monitors, new pacemaker and implantable cardioverter defibrillators (ICD), and other EP lab technologies.
Find links and more news from the Heart Rhythm Society 2017 late-breaking electrophysiology trials.
Bruce Wilkoff, M.D., director of cardiac pacing and tachyarrhythmia devices at Cleveland Clinic, discusses advancements in lead management technology at the 2017 Heart Rhythm Society (HRS) annual meeting. Read the article “ Study Shows Occlusion Balloon Saves Lives During Lead Extraction.” Read the articles “Advances in Transvenous Lead Extraction,” and “Strategies, New Technologies Aid Lead Management.”
Vivek Reddy, M.D., director of cardiac arrhythmia services and professor of medicine, cardiology, Mount Sinai Hospital, N.Y., discusses advancements in leadless pacemaker technology at the 2017 Heart Rhythm Society (HRS) annual meeting. He explains the pros and cons of leadless pacemaker technology as it stands today. One of the issues is that leadless pacing is currently limited to single chamber pacing, while most patients require dual chamber pacing. Reddy discusses the technology that currently has U.S. FDA market clearance, the Medtronic Micra, and new technologies in development to overcome the limitations the Micra.
Read the related articles “Leadless Endocardial CRT Pacing Effective for Heart Failure Patients,” and “New Data Showcases Long-term Benefits of Nanostim Leadless Pacemaker.”
This video, provided by ERB, demonstrates the function and implantation of the WiSE CRT (Wireless Stimulation Endocardially for cardiac resynchronization therapy) leadless CRT pacing system for heart failure patients. Read the article “Leadless Endocardial CRT Pacing Effective for Heart Failure Patients.”
Hugh Calkins, M.D., FACC, FAHA, FHRS, director of cardiac arrhythmia services and professor of medicine at Johns Hopkins Hospital, discusses advancements in AF ablation at the 2017 Heart Rhythm Society (HRS) annual meeting. Read about the new AF expert consensus statement released at HRS 2017.
Related Content:
VIDEO: Current State of Atrial Fibrillation Ablation Technologies — Interview with Hugh Calkins, M.D.,
New Technologies to Improve Atrial Fibrillation Ablation
Landmark CABANA Trial Confirms Ablation Equal To or Superior to Drug Therapy
VIDEO: Cryoballoon Ablation Best Practice Guidelines — Wilber Su, M.D.
This video, provided by Spectranetics, demonstrates how to deploy the Bridge Occlusion Balloon used to seal accidental tears in the superior vena cava (SVC) during EP device lead extraction procedures. A review of post-market release data shown it can bridge the patient to open surgical repair and significantly reduce the usual 50 percent mortality associated with this complication. Read the article about the late-breaking clinical trial presentation from Heart Rhythm 2017.
This video, provided by Medtronic, demonstrates the implantation of Micra transcatheter pacing system (TPS). The device is a leadless pacemaker for single chamber pacing and eliminated the need for a surgical pocket or transvenous leads. The design concept is to simplify pacemaker implantation and eliminate the need for leads, which open the patient up to additional device and infection complications.
Watch the VIDEO “Current State of Leadless Pacemaker Technology,” an interview with Vivek Reddy, M.D., at the Heart Rhythm Society 2017 meeting.
Related Micra Leadless Pacemaker Content:
FDA Clears Medtronic Micra AV to Treat AV Block
New Algorithms in Medtronic Micra Pacemaker May Improve Synchrony and Cardiac Function in AV Block
FDA Approves World's Smallest Pacemaker for U.S. Patients
Safety, Performance of the World's Smallest Pacemaker Reinforced in Real-world Patients
One-Year Results for Micra TPS Pacemaker Trial Presented at ESC 2016
Leadless Pacemaker Gains Medicare Reimbursement
Wireless and Leadless Pacemaker Being Developed by Texas Heart Institute With Federal Grant
A discussion with Heart Rhythm Society (HRS) President Michael Gold, M.D., Ph.D., director of cardiology and associate dean at the Medical University of South Carolina (MUSC), at the American College of Cardiology 2017 annual meeting. He discusses advances to treat atrial fibrillation, new ablation and electro mapping technologies and innovations in implantable devices.
Read the article "What is New in Electrophysiology Technologies."
DAIC Editor Dave Fornell takes a tour of some of the interesting new technologies from the vendor booths on the expo floor at the 2017 meeting of the American College of Cardiology (ACC). For key technology trends at ACC.17, watch the VIDEO "Cardiac Technology Trends at ACC.17."
This video, provided by Medtronic, demonstrates the CardioInsight electro-anatomical mapping system. It was cleared by the FDA in early 2017. It uses an ECG lead vest to noninvasively create a 3-D electrophysiology (EP) electro-map of the heart to help identify arrhythmia and plan catheter ablation procedures. Read the related article on the technology.
William Abraham, M.D., FACC, discusses advances in heart failure device treatment technologies at the Transcatheter Cardiovascular Therapeutics (TCT) 2016 annual meeting. He is director of the division of cardiovascular medicine and a professor of internal medicine, physiology and cell biology at The Ohio State University Wexner Medical Center. He also served as principal investigator of the CHAMPION Trial for the CardioMEMS device. Read the article "Reducing Heart Failure Readmissions."
This video, provided by Zoll, demonstrates how cardiologists can explain sudden cardiac death to patients. It is critical to educate patients who are at risk of sudden cardiac arrest (SCA), so Rahul Doshi, M.D., director of electrophysiology, associate professor of clinical medicine, Keck Medical Center of University of Southern California, explains sudden cardiac death (SCD) to his patients using a simple illustration. Visit SCDFacts.org for additional resources for you and your team to support the SCD conversation with your patients. One in five post-AMI patients have been shown to be at high risk of dying after PCI.1 The majority of mortality in AMI patients post-PCI occurs in the first three months — one out of every 10 high-risk patients die, with about 60 percent of this mortality due to SCD.[1,2]
2. Stone G, et al. Prevention of Sudden Cardiac Arrest Post PTCA in High-Risk Patients. Medscape. April 2011. https://www.medscape.org/viewarticle/741503_transcript
With quality of care and cost efficiency at the top of your mind, there is no room in your hospital for waste from high-value supplies. However, managing your critical supplies in the cath lab can be a challenge. How can you get the supply waste in your facility under control? Watch this short video to learn how an automated inventory management solution could help you reduce the waste in your hospital while improving your total cost of care. For more information visit www.cardinalhealth.com.
With bundled payments putting increased pressure on hospitals to manage supply costs while providing quality patient care, there is no room in your cath lab for high-value medical device waste. An automated inventory management solution could help you find and reduce the waste hiding in your supply chain while helping to improve your total cost of care. Visit cardinalhealth.com/CIMS.
This video, provided by Respicardia, demonstrates the Remede System is a pacemaker-like implantable device. It is designed to improve cardiovascular health by restoring natural breathing during sleep in patients with central sleep apnea. In this video from The Ohio State University, William Abraham, M.D., director of the Division of Cardiovascular Medicine at The Ohio State University Wexner Medical Center, explains how the technology works and highlights one patient case involved in a recent study of the device.
Pacemakers, stents and bandages — keeping tracking of what is on hand and accurately capturing charges can be a challenge. What if you could track, manage and analyze your cath lab inventory – low cost to high value – to reveal powerful business intelligence and shine a light on new savings and revenue capture?
David Holmes, M.D., professor of medicine, Mayo Clinic College of Medicine and consultant, Department of Internal Medicine, Division of Cardiovascular Diseases, shares details from his presentations at American College of Cardiology (ACC) 2016 regarding the Watchman left atrial appendage (LAA) occluder. The big news from ACC regarding LAA occluders was his presentation of real-life experience of centers using the Watchman outside of trials.
Managing inventory in the procedural area is often a challenge that creates substantial waste and inefficiency that impacts your bottom line. Understanding how to overcome the top barriers allows you to spend more time on patient care and less time on tackling your supplies. Learn how you can manage inventory more effectively with Cardinal Health Inventory Management Solutions. For more information visit cardinalhealth.com/cims.
Learn about the trends and new technology in electrophysiology (EP) in an interview with David Wilber, M.D., editor of JACC's Clinical EP, and medical director, clinical electrophysiology, director, Cardiovascular Institute, co-director, Cardiovascular Research Institute, Loyola University Medical Center, Chicago.
DAIC Editor Dave Fornell shares his picks of the most interesting new devices and advances in cardiovascular technology shown on the expo floor at the 2015 American College of Cardiology (ACC) meeting.
Paul Keall, Ph.D., FAAP, professor and NHMRC Australian Fellow at Radiation Physics Laboratory at the University of Sydney, Sydney Medical School, discusses radiosurgery beyond cancer.
DAIC Editor Dave Fornell offers his choices of the most innovative new cardiovascular technologies shown on the expo floor at the American College of Cardiology (ACC) 2014 meeting.
DAIC Editor Dave Fornell offers his choices of the most innovative new cardiovascular technologies discussed in sessions and shown on the expo floor at the Transcatheter Cardiovascular Therapeutics (TCT) 2013 meeting.
This video, provided by Abbott/St. Jude Medical, demonstrates how the Nanostim leadless pacemaker is delivered via catheter. This eliminates the need for open surgical procedures. It is anchored into the myocardium at apex of the right ventricle. Watch the VIDEO “Current State of Leadless Pacemaker Technology,” an interview with Vivek Reddy, M.D.
McKesson highlighted enhancements to its cardiovascular information system (CVIS) version 13. The system offers new modules for electrophysiology, inventory control and aids interoperability for better change capture and data mining. For more information, visit www.allaboutCVIS.com
To help hospitals upgrade their electrophysiology (EP) reporting systems and integrate EP into cardiovascular and hospital reporting systems, Michael Mirro, M.D., FACC, medical director, cardiology/electrophysiology, Parkview Physicians Group, Ft. Wayne, Ind., offers suggestions of what to look for in systems. He spoke about cardiovascular/EP IT integration and the impact of healthcare reforms during the Heart Rhythm Society (HRS) 2012 scientific sessions.
Data was presented during the Heart Rhythm Society (HRS) 2012 scientific sessions that showed the Riata ICD lead had a higher malfunction rate than comparable leads. The results from "Independent Multicenter Study of Riata and Riata ST Implantable Cardioverter Defibrillator (ICD) Leads" were presented by Raed H. Abdelhadi, M.D., FACC, a cardiologist and electrophysiologist researcher at the Minneapolis Heart Institute Foundation.
Diagnostic and Interventional Cardiology Editor Dave Fornell shows some of the most innovative new technology displayed on the show floor at the Heart Rhythm Society (HRS) annual scientific sessions. The tour includes new devices and trends in electrophysiology. For more information: www.DIcardiology.com
Heart Rhythm Society (HRS) President-Elect Hugh Calkins, M.D., FACC, director, cardiac arrhythmia services and EP lab, Johns Hopkins University, Baltimore, Md., offers an overview of trends and top news in electrophysiology at the HRS 2012 scientific sessions. For more information: www.DIcardiology.com
Pivitol trial data regarding the safety and efficacy of the Cameron Health Subcutaneous Implantable Cardioverter Defibrillator (S-ICD) system was very positive. The system is currently pending FDA approval and would be the only subcutaneous lead electrophysiology device cleared for use in the United States. The system eliminates the need for venous leads and intra-cardiac securement, greatly simplifying the implant procedure. Data from Cameron Health's IDE trial was presented by Martin Burke, DO, FACC, FACOI, FRCP, director, Heart Rhythm Center, University of Chicago, at the Heart Rhythm Society (HRS) 2012 scientific sessions. For more information: www.DIcardiology.com
Implantable cardioverter defibrillators (ICDs) are safe in patients who participate in sports, according to a study presented during Heart Rhythm Society (HRS) 2012. Rachel Lampert, M.D., associate professor of medicine, section of cardiovascular medicine, Yale School of Medicine, explains the study findings during a press briefing at HRS. For more information: www.DIcardiology.com
Siemens' AcuNav V 3-D intracardiac echo (ICE) catheter offers detailed, live 3-D images of the interior of the heart. This video shows an example of the catheter imaging the pulmonary vein. The technology may play a role in better guiding transcatheter electrophysiology (EP) ablation procedures. The technology was shown as a work-in-progress during ACC 2012.