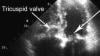
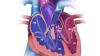
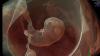
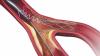
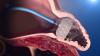
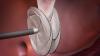
Rebecca Hahn, M.D., professor of medicine and director of interventional echocardiography, Columbia University Medical Center, New York, explains that techniques for imaging the tricuspid valve, the advanced study of its disease etiology and the rapid development of transcatheter devices to treat tricuspid valve disease are all developing together. She spoke on these topics during sessions at both the American Society of Echocardiography (ASE) 2018 meeting and at the Transcatheter Valve Therapies (TVT) conference in June.
Related Transcatheter Tricuspid Valve Content:
Recent Advances in Transcatheter Valve Technology
VIDEO: Mitral and Tricuspid Valve Repair Technologies — interview with Azeem Latib, M.D.
VIDEO: Transcatheter Tricuspid Valve Repair and Replacement Technologies — interview with Rebecca Hahn, M.D.
Minneapolis Heart Institute Foundation Enrolls First Patient in TRILUMINATE Tricuspid Repair Trial
Positive, Sustained Improvement at One Year in SCOUT I Transcatheter Tricuspid Repair Trial